Tay-Sachs disease is a rare genetic disorder that is typically constrained to certain ethnic populations. It is characterized by the destruction of nerve cells in the central nervous system and can lead to blindness, deafness, and the loss of mental and physical functions.
Tay-Sachs disease is a rare inherited disorder that progressively destroys nerve cells (neurons) in the brain and spinal cord. The most common form of Tay-Sachs disease becomes apparent in infancy. Infants with this disorder typically appear normal until the age of 3 to 6 months, when their development slows and muscles used for movement weaken. Tay-Sachs disease is a rare genetic disorder which causes progressive nerve destruction, leading to a variety of symptoms. Learn more about it.
Tay-Sachs is a recessive autosomal disorder, meaning that it is a disease inherited from one’s parents. The disease is associated with mutations in the HEXA gene of which there are more than 100 variations. Depending on which mutations are paired, the disease can take vastly different forms, causing illness during infancy, childhood, or adulthood.
Death usually occurs in children within a few years of the first symptoms, as the destruction of nerve cells leaves them unable to move or even swallow. In the advanced stages, children will become increasingly prone to infection. Most die from complications of pneumonia or other respiratory infections.
While rare in the general population, Tay-Sachs is seen more frequently in Ashkenazi Jews, Cajuns of southern Louisiana, French Canadians of Southern Quebec, and Irish Americans.
How It Causes Illness
The HEXA gene provides instructions for making a type of enzyme known as hexosaminidase A. This enzyme is responsible for breaking down a fatty substance known as GM2 ganglioside.
In persons with Tay-Sachs disease, hexosaminidase A no longer functions as it should. Without the means to break down these fats, toxic levels begin to accumulate in the nerve cells of the brain and spinal cord, eventually destroying them and leading to the symptoms of the disease.
Infantile Tay-Sachs Disease
The signs and symptoms of Tay-Sachs disease vary by when the symptoms first develop.
The most common type is infantile Tay-Sachs disease, the first signs of which will appear between the ages of three and six months. This is the time when parents will start to notice that their child’s development and movements have markedly slowed.
By this stage, the child may be unable to turn over or sit up. If the child is older, he or she may have difficulty crawling or lifting an arm. They will also exhibit an exaggerated, startled reaction to loud noises and may appear listless or lethargic. From there, the deterioration of mental and physical functions is usually relentless and profound.
Symptoms typically include:
- Seizures
- Vision loss and blindness
- Hearing loss and deafness
- Drooling and difficulty swallowing (dysphagia)
- Muscle stiffness and spasticity
- Muscle atrophy
- Loss of intellectual and cognitive function
- Paralysis
- Death
Death, even with the best of medical care, usually occurs before the age of four.
Less Common Forms
While infantile Tay-Sachs is the predominant form of the disease, there are less common types seen in children and adults. The timing of the onset of the disease is largely related to the combination of genes inherited from the parents.
Of the two less common forms:
- Juvenile Tay-Sachs disease usually manifests with symptoms anywhere from two to 10 years of age. As with infantile Tay-Sachs, the progression of the disease is unremitting, leading to deterioration of cognitive skills, speech, and muscles coordination as well as dysphagia and spasticity. Death usually occurs between five and 15 years.
- Late-onset Tay-Sachs disease (also known as adult Tay-Sachs) is extremely rare and will usually develop in adults between 30 and 40. Unlike the other forms of the disease, it is usually not fatal and will stop progressing after a certain amount of time. During this time, a person may experience a decline in mental skills, unsteadiness, speech difficulties, dysphagia, spasticity, and even a schizophrenia-like psychosis. Persons with late-onset Tay Sachs can often become permanently disabled and require a full-time wheelchair.
Genetics and Inheritance
As with any autosomal recessive disorder, Tay-Sachs occurs when two parents who don’t have the disease each contribute a recessive gene to their offspring. The parents are considered 'carriers' because they each have one dominant (normal) copy of the gene and one recessive (mutated) copy of the gene. It is only when a person has two recessive genes that Tay-Sachs can occur.
If both parents are carriers, a child has a 25 percent chance of inhering two recessive genes (and getting Tay-Sachs), a 50 percent chance of getting one dominant and one recessive gene (and becoming a carrier), and a 25 percent chance of getting two dominant genes (and remaining unaffected).
Because there are well over 100 variations of the HEXA mutation, different recessive combinations can end up meaning vastly different things. In some cases, a combination will confer to earlier onset and faster disease progression and, in other cases, later onset and slower disease progression.

While scientists are closer to understanding which combinations relate to which forms of the disease, there are still large gaps in our genetic understanding of Tay-Sachs in all of its forms.
Risk
As rare as Tay-Sachs is in the general population—occurring in around one of every 320,000 births—there are certain populations where the risk is considerably higher.
The risk is largely constrained to so-called 'founder populations' in which groups can trace the roots of the disease back to a specific, common ancestor. Due to the lack of genetic diversity within these groups, certain mutations are passed more readily to offspring, resulting in higher rates of autosomal diseases.
With Tay-Sachs, we see this with four specific groups:
- Ashkenazi Jews have the highest risk of Tay-Sachs, with one of every 30 persons being a carrier. The specific type of mutation seen within the population (called 1278insTATC) leads to infantile Tay-Sachs disease. In the United States, one of every 3,500 Ashkenazi babies will be affected.
- Cajuns in southern Louisiana are also affected by the same mutation and have more or less the same level of risk. Scientists have been able to trace the ancestry of carriers back to the 18th century to a single founder couple in France who were not Jewish.
- French Canadians in Quebec have the same risk as Cajuns and Ashkenazi Jews but are affected by two entirely unrelated mutations. Within this population, one or both mutations are believed to have been brought from Normandy or Perche to the colony of New France sometime around the 17th century.
- Irish Americans are less commonly affected but still have around a one in 50 chance of being a carrier. The risk appears to be greatest among couples who have at least three Irish grandparents between them.
Diagnosis
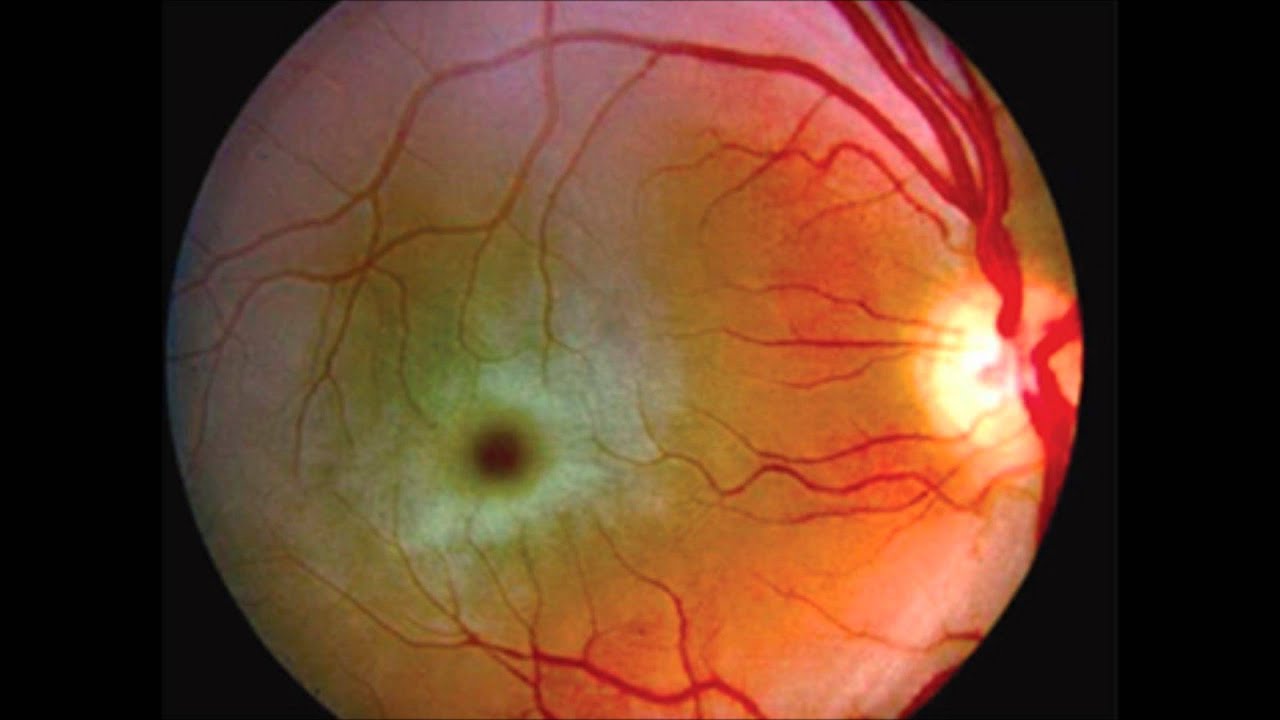
Beyond the motor and cognitive symptoms of the disease, one of the tell-tale signs in children is an eye abnormality called a 'cherry spot.' The condition, characterized by an oval, red discoloration on the retina, is easily spotted during a routine eye exam. The cherry spot is seen in all infants with Tay-Sachs disease as well as some children. It is not seen in adults.
Based on the family history and the appearance of symptoms, the doctor will confirm the diagnosis by ordering blood tests to assess the levels of hexosaminidase A, which will either be low or non-existent. If there is any doubt about the diagnosis, the doctor can perform a genetic test to confirm the HEXA mutation.
Treatment
There is no cure for Tay-Sachs disease. Treatment is primarily directed at the management of symptoms, which may include:
- Anti-seizure medications such as gabapentin or lamotrigine
- Antibiotics to prevent or treat aspiration pneumonia
- Chest physiotherapy to treat respiratory congestion
- Physical therapy to keep the joints flexible
- Medications such as trihexyphenidyl or glycopyrrolate to control production of saliva and prevent drooling
- Using baby bottles designed for children with cleft palates to aid in swallowing
- Muscle relaxants like diazepam to treat stiffness and cramping
- Feeding tubes either delivered through the nose (nasogastric) or surgically implanted into the stomach (percutaneous endoscopic gastrostomy, or PEG tube)
While gene therapy and enzyme replacement therapy research are being explored as a means to cure or slow the progression Tay-Sachs disease, most all are in the very early stage of research.
Prevention
Ultimately, the only way to prevent Tay-Sachs is to identify couples who are at high risk and aid them in making the appropriate reproductive choices. Depending on the situation, an intervention may take place either before or during a pregnancy. In some cases, there may be ethical or moral dilemmas to consider.
Among the options:
- During a pregnancy, amniocentesis or chorionic villus sampling (CVS) can be used to retrieve fetal cells for genetic testing. Couples can then decide whether to have an abortion based on the results.
- Couples who are undergoing in vitro fertilization (IVF) can have the fertilized eggs genetically tested before implantation. By doing so, the couple can ensure that only healthy embryos are transferred to the womb. While effective, IVF is a very costly option.
- In orthodox Jewish communities, a group called Dor Yeshorim conducts anonymous screenings for Tay-Sachs disease in high school students. Persons who undergo the testing are given a six-digit identification code. When they find a potential mate, the couple can call a hotline and use their ID numbers assess their 'compatibility.' If both are carriers, they would be designated as 'incompatible.'
A Word From Verywell
If faced with a positive result for Tay-Sachs disease—either as a carrier or a parent—it is important to speak with a specialist physician to fully understand what the diagnosis means and what your options are. There are no wrong or right choices, just personal ones for which you and partner have every right to confidentiality and respect.
- Bley, A.; Giannikopoulos, O.; Hayden D. et al. 'Natural history of infantile G(M2) gangliosidosis.' Pediatrics. 2011; 128:e1233-41. DOI: 10.1542/peds.2011-0078.
- Hall, P.; Minnich, S.; Teigen, C. et al. 'Diagnosing lysosomal storage disorders: the GM2 gangliosidoses.' Curr Protoc Hum Genet. 2014; 83:17.161-8. DOI: 10.1002/0471142905.hg1716s83.
- National Institutes of Health. 'Tay-Sachs Disease.' Gen Home Ref. Bethesda, Maryland; updated January 23, 2018.
- Schneider, A.; Nakagawa, S.; Keep, R. et al. 'Population-based Tay-Sachs screening among Ashkenazi Jewish young adults in the 21st century: hexosaminidase A enzyme assay is essential for accurate testing.' Am J Med Genet A. 2009; 149A:2444-7. DOI: 10.1002/ajmg.a.33085.
- Steiner K.; Brenck, J.; Goericke, S. et al. 'Cerebellar atrophy and muscle weakness: late-onset Tay-Sachs disease outside Jewish populations.' BMJ Case Rep. 2016; 2016: bcr2016214634. DOI: 10.1136/bcr-2016-214634.
| Tay–Sachs disease | |
|---|---|
| Other names | GM2 gangliosidosis, hexosaminidase A deficiency[1] |
| Cherry-red spot as seen in the retina in Tay–Sachs disease. The fovea's center appears bright red because it is surrounded by a whiter than usual area. | |
| Specialty | Medical genetics |
| Symptoms | Initially: Decreased ability to turn over, sit, or crawl[1] Later: Seizures, hearing loss, inability to move[1] |
| Usual onset | Three to six months of age[1] |
| Duration | Long term[2] |
| Types | Infantile, juvenile, late-onset[2] |
| Causes | Genetic (autosomal recessive)[1] |
| Diagnostic method | Testing blood hexosaminidase A levels, genetic testing[2] |
| Differential diagnosis | Sandhoff disease, Leigh syndrome, neuronal ceroid lipofuscinoses[2] |
| Treatment | Supportive care, psychosocial support[2] |
| Prognosis | Death often occurs in early childhood[1] |
| Frequency | Rare in the general population[1] |
Tay–Sachs disease is a genetic disorder that results in the destruction of nerve cells in the brain and spinal cord.[1] The most common type, known as infantile Tay–Sachs disease, becomes apparent around three to six months of age with the baby losing the ability to turn over, sit, or crawl.[1] This is then followed by seizures, hearing loss, and inability to move.[1] Death usually occurs in early childhood.[1] Less commonly the disease may occur in later childhood or adulthood.[1] These forms are generally milder in nature.[1]
Tay–Sachs disease is caused by a genetic mutation in the HEXA gene on chromosome 15.[1] It is inherited from a person's parents in an autosomal recessive manner.[1] The mutation results in problems with an enzyme called beta-hexosaminidase A which results in the buildup of the molecule GM2 ganglioside within cells, leading to toxicity.[1] Diagnosis is by measuring the blood hexosaminidase A level or genetic testing.[2] It is a type of GM2 gangliosidosis and a type of sphingolipidosis.[3]
The treatment of Tay–Sachs disease is supportive in nature.[2] This may involve multiple specialities as well as psychosocial support for the family.[2] The disease is rare in the general population.[1] In Ashkenazi Jews, French Canadians of southeastern Quebec, and Cajuns of southern Louisiana, the condition is more common.[2][1] Approximately 1 in 3,600 Ashkenazi Jews at birth are affected.[2]
The disease is named after Waren Tay, who in 1881 first described a symptomatic red spot on the retina of the eye; and Bernard Sachs, who described in 1887 the cellular changes and noted an increased rate of disease in Ashkenazi Jews.[4] Carriers of a single Tay–Sachs allele are typically normal.[2] It has been hypothesized that being a carrier may confer protection from another condition such as tuberculosis, explaining the persistence of the allele in certain populations.[5] Researchers are looking at gene therapy or enzyme replacement therapy as possible treatments.[2]
- 1Signs and symptoms
- 11Research directions
Signs and symptoms[edit]
Tay–Sachs disease is typically first noticed in infants around 6 months old displaying an abnormally strong response to sudden noises or other stimuli, known as the 'startle response'. There may also be listlessness or muscle stiffness (hypertonia). The disease is classified into several forms, which are differentiated based on the onset age of neurologicalsymptoms.[6][7]
Infantile[edit]
Infants with Tay–Sachs disease appear to develop normally for the first six months after birth. Then, as neurons become distended with gangliosides, a relentless deterioration of mental and physical abilities begins. The child may become blind, deaf, unable to swallow, atrophied, and paralytic. Death usually occurs before the age of four.[6]
Juvenile[edit]
Juvenile Tay–Sachs disease is rarer than other forms of Tay–Sachs, and usually is initially seen in children between two and ten years old. People with Tay–Sachs disease develop cognitive and motor skill deterioration, dysarthria, dysphagia, ataxia, and spasticity.[8] Death usually occurs between the age of five to fifteen years.[9]
Late-onset[edit]
A rare form of this disease, known as Adult-Onset or Late-Onset Tay–Sachs disease, usually has its first symptoms during the 30s or 40s. In contrast to the other forms, late-onset Tay–Sachs disease is usually not fatal as the effects can stop progressing. It is frequently misdiagnosed. It is characterized by unsteadiness of gait and progressive neurological deterioration. Symptoms of late-onset Tay–Sachs – which typically begin to be seen in adolescence or early adulthood – include speech and swallowing difficulties, unsteadiness of gait, spasticity, cognitive decline, and psychiatric illness, particularly a schizophrenia-like psychosis.[10] People with late-onset Tay–Sachs may become full-time wheelchair users in adulthood.
Until the 1970s and 1980s, when the disease's molecular genetics became known, the juvenile and adult forms of the disease were not always recognized as variants of Tay–Sachs disease. Post-infantile Tay–Sachs was often misdiagnosed as another neurological disorder, such as Friedreich's ataxia.[11]
Genetics[edit]
Tay–Sachs disease is inherited in the autosomal recessive pattern, depicted above.
The HEXA gene is located on the long (q) arm of human chromosome 15, between positions 23 and 24.
Tay–Sachs disease is an autosomal recessive genetic disorder, meaning that when both parents are carriers, there is a 25% risk of giving birth to an affected child with each pregnancy. The affected child would have received a mutated copy of the gene from each parent.[6] Tay–Sachs results from mutations in the HEXAgene on chromosome 15, which encodes the alpha-subunit of beta-N-acetylhexosaminidase A, a lysosomalenzyme. By 2000, more than 100 different mutations had been identified in the human HEXA gene.[12] These mutations have included single base insertions and deletions, splice phase mutations, missense mutations, and other more complex patterns. Each of these mutations alters the gene's protein product (i.e., the enzyme), sometimes severely inhibiting its function.[13] In recent years, population studies and pedigree analysis have shown how such mutations arise and spread within small founder populations. Initial research focused on several such founder populations:
- Ashkenazi Jews. A four base pair insertion in exon 11 (1278insTATC) results in an altered reading frame for the HEXA gene. This mutation is the most prevalent mutation in the Ashkenazi Jewish population, and leads to the infantile form of Tay–Sachs disease.[14]
- Cajuns. The same 1278insTATC mutation found among Ashkenazi Jews occurs in the Cajun population of southern Louisiana. Researchers have traced the ancestry of carriers from Louisiana families back to a single founder couple – not known to be Jewish – who lived in France in the 18th century.[15]
- French Canadians. Two mutations, unrelated to the Ashkenazi/Cajun mutation, are absent in France but common among French Canadians living in eastern Quebec and Acadians from the Province of New Brunswick. Pedigree analysis suggests the mutations were uncommon before the late 17th century.[16][17]
In the 1960s and early 1970s, when the biochemical basis of Tay–Sachs disease was first becoming known, no mutations had been sequenced directly for genetic diseases. Researchers of that era did not yet know how common polymorphisms would prove to be. The 'Jewish Fur Trader Hypothesis,' with its implication that a single mutation must have spread from one population into another, reflected the knowledge at the time. Subsequent research, however, has proven that a large variety of different HEXA mutations can cause the disease. Because Tay–Sachs was one of the first genetic disorders for which widespread genetic screening was possible, it is one of the first genetic disorders in which the prevalence of compound heterozygosity has been demonstrated.[18]
Compound heterozygosity ultimately explains the disease's variability, including the late-onset forms. The disease can potentially result from the inheritance of two unrelated mutations in the HEXA gene, one from each parent. Classic infantile Tay–Sachs disease results when a child has inherited mutations from both parents that completely stop the biodegradation of gangliosides. Late onset forms occur due to the diverse mutation base – people with Tay–Sachs disease may technically be heterozygotes, with two differing HEXA mutations that both inactivate, alter, or inhibit enzyme activity. When a patient has at least one HEXA copy that still enables some level of hexosaminidase A activity, a later onset disease form occurs. When disease occurs because of two unrelated mutations, the patient is said to be a compound heterozygote.[19]
Heterozygous carriers (individuals who inherit one mutant allele) show abnormal enzyme activity but manifest no disease symptoms. This phenomenon is called dominance; the biochemical reason for wild-type alleles' dominance over nonfunctional mutant alleles in inborn errors of metabolism comes from how enzymes function. Enzymes are proteincatalysts for chemical reactions; as catalysts, they speed up reactions without being used up in the process, so only small enzyme quantities are required to carry out a reaction. Someone homozygous for a nonfunctional mutation in the enzyme-encoding gene has little or no enzyme activity, so will manifest the abnormal phenotype. A heterozygote (heterozygous individual) has at least half of the normal enzyme activity level, due to expression of the wild-type allele. This level is normally enough to enable normal function and thus prevent phenotypic expression.[20]
Pathophysiology[edit]
Tay–Sachs disease is caused by insufficient activity of the enzyme hexosaminidase A. Hexosaminidase A is a vital hydrolytic enzyme, found in the lysosomes, that breaks down sphingolipids. When hexosaminidase A is no longer functioning properly, the lipids accumulate in the brain and interfere with normal biological processes. Hexosaminidase A specifically breaks down fatty acid derivatives called gangliosides; these are made and biodegraded rapidly in early life as the brain develops. Patients with and carriers of Tay–Sachs can be identified by a simple blood test that measures hexosaminidase A activity.[6]
The hydrolysis of GM2-ganglioside requires three proteins. Two of them are subunits of hexosaminidase A; the third is a small glycolipid transport protein, the GM2 activator protein (GM2A), which acts as a substrate-specific cofactor for the enzyme. Deficiency in any one of these proteins leads to ganglioside storage, primarily in the lysosomes of neurons. Tay–Sachs disease (along with AB-variant GM2-gangliosidosis and Sandhoff disease) occurs because a mutation inherited from both parents deactivates or inhibits this process. Most Tay–Sachs mutations probably do not directly affect protein functional elements (e.g., the active site). Instead, they cause incorrect folding (disrupting function) or disable intracellular transport.[21]
Diagnosis[edit]
In patients with a clinical suspicion for Tay–Sachs disease, with any age of onset, the initial testing involves an enzyme assay to measure the activity of hexosaminidase in serum, fibroblasts, or leukocytes. Total hexosaminidase enzyme activity is decreased in individuals with Tay–Sachs as is the percentage of hexosaminidase A. After confirmation of decreased enzyme activity in an individual, confirmation by molecular analysis can be pursued.[22] All patients with infantile onset Tay–Sachs disease have a 'cherry red' macula in the retina, easily observable by a physician using an ophthalmoscope.[6][23] This red spot is a retinal area that appears red because of gangliosides in the surrounding retinal ganglion cells. The choroidal circulation is showing through 'red' in this foveal region where all retinal ganglion cells are pushed aside to increase visual acuity. Thus, this cherry-red spot is the only normal part of the retina; it shows up in contrast to the rest of the retina. Microscopic analysis of the retinal neurons shows they are distended from excess ganglioside storage.[24] Unlike other lysosomal storage diseases (e.g., Gaucher disease, Niemann–Pick disease, and Sandhoff disease), hepatosplenomegaly (liver and spleen enlargement) is not seen in Tay–Sachs.[25]
Prevention[edit]
Three main approaches have been used to prevent or reduce the incidence of Tay–Sachs:
- Prenatal diagnosis. If both parents are identified as carriers, prenatal genetic testing can determine whether the fetus has inherited a defective gene copy from both parents.[26]Chorionic villus sampling (CVS), the most common form of prenatal diagnosis, can be performed between 10 and 14 weeks of gestation. Amniocentesis is usually performed at 15–18 weeks. These procedures have risks of miscarriage of 1% or less.[27][28]
- Preimplantation genetic diagnosis. By retrieving the mother's eggs for in vitro fertilization, it is possible to test the embryo for the disorder prior to implantation. Healthy embryos are then selected and transferred into the mother's womb, while unhealthy embryos are discarded. In addition to Tay–Sachs disease, preimplantation genetic diagnosis has been used to prevent cystic fibrosis and sickle cell anemia among other genetic disorders.[29]
- Mate selection. In Orthodox Jewish circles, the organization Dor Yeshorim carries out an anonymous screening program so that carriers for Tay–Sachs and other genetic disorders can avoid marrying each other.[30]
Management[edit]
As of 2010 there was no treatment that addressed the cause of Tay–Sachs disease or could slow its progression; people receive supportive care to ease the symptoms and extend life by reducing the chance of contracting infections.[31] Infants are given feeding tubes when they can no longer swallow.[32] In late-onset Tay–Sachs, medication (e.g., lithium for depression) can sometimes control psychiatric symptoms and seizures, although some medications (e.g., tricyclic antidepressants, phenothiazines, haloperidol, and risperidone) are associated with significant adverse effects.[19][33]
Outcomes[edit]
As of 2010, even with the best care, children with infantile Tay–Sachs disease usually die by the age of 4. Children with the juvenile form are likely to die from the ages 5–15, while those with the adult form will probably not be affected.[31]
Epidemiology[edit]
Founder effects occur when a small number of individuals from a larger population establish a new population. In this illustration, the original population is on the left with three possible founder populations on the right. Two of the three founder populations are genetically distinct from the original population.
Ashkenazi Jews have a high incidence of Tay–Sachs and other lipid storage diseases. In the United States, about 1 in 27 to 1 in 30 Ashkenazi Jews is a recessive carrier. The disease incidence is about 1 in every 3,500 newborn among Ashkenazi Jews.[34]French Canadians and the Cajun community of Louisiana have an occurrence similar to the Ashkenazi Jews. Irish Americans have a 1 in 50 chance of being a carrier.[citation needed] In the general population, the incidence of carriers as heterozygotes is about 1 in 300.[7] The incidence is approximately 1 in 320,000 newborns in the general population in United States.[35]
Three general classes of theories have been proposed to explain the high frequency of Tay–Sachs carriers in the Ashkenazi Jewish population:
- Heterozygote advantage.[36] When applied to a particular allele, this theory posits that mutation carriers have a selective advantage, perhaps in a particular environment.[37]
- Reproductive compensation. Parents who lose a child because of disease tend to 'compensate' by having additional children to replace them. This phenomenon may maintain and possibly even increase the incidence of autosomal recessive disease.[38]
- Founder effect. This hypothesis states that the high incidence of the 1278insTATC chromosomes[37] is the result of an elevated allele frequency[36] that existed by chance in an early founder population.[37]
Tay–Sachs disease was one of the first genetic disorders for which epidemiology was studied using molecular data. Studies of Tay–Sachs mutations using new molecular techniques such as linkage disequilibrium and coalescence analysis have brought an emerging consensus among researchers supporting the founder effect theory.[37][39][40]
History[edit]
Waren Tay and Bernard Sachs, two physicians, described the disease's progression and provided differential diagnostic criteria to distinguish it from other neurological disorders with similar symptoms.
Both Tay and Sachs reported their first cases among Ashkenazi Jewish families. Tay reported his observations in 1881 in the first volume of the proceedings of the British Ophthalmological Society, of which he was a founding member.[41] By 1884, he had seen three cases in a single family. Years later, Bernard Sachs, an American neurologist, reported similar findings when he reported a case of 'arrested cerebral development' to other New York Neurological Society members.[42][43]
Sachs, who recognized that the disease had a familial basis, proposed that the disease should be called amaurotic familial idiocy. However, its genetic basis was still poorly understood. Although Gregor Mendel had published his article on the genetics of peas in 1865, Mendel's paper was largely forgotten for more than a generation – not rediscovered by other scientists until 1899. Thus, the Mendelian model for explaining Tay–Sachs was unavailable to scientists and doctors of the time. The first edition of the Jewish Encyclopedia, published in 12 volumes between 1901 and 1906, described what was then known about the disease:[44]
It is a curious fact that amaurotic family idiocy, a rare and fatal disease of children, occurs mostly among Jews. The largest number of cases has been observed in the United States—over thirty in number. It was at first thought that this was an exclusively Jewish disease because most of the cases at first reported were between Russian and Polish Jews; but recently there have been reported cases occurring in non-Jewish children. The chief characteristics of the disease are progressive mental and physical enfeeblement; weakness and paralysis of all the extremities; and marasmus, associated with symmetrical changes in the macula lutea. On investigation of the reported cases, they found that neither consanguinity nor syphilitic, alcoholic, or nervous antecedents in the family history are factors in the etiology of the disease. No preventive measures have as yet been discovered, and no treatment has been of benefit, all the cases having terminated fatally.
Jewish immigration to the United States peaked in the period 1880–1924, with the immigrants arriving from Russia and countries in Eastern Europe; this was also a period of nativism (hostility to immigrants) in the United States. Opponents of immigration often questioned whether immigrants from southern and eastern Europe could be assimilated into American society. Reports of Tay–Sachs disease contributed to a perception among nativists that Jews were an inferior race.[43]
In 1969, Shintaro Okada and John S. O'Brien showed that Tay–Sachs disease was caused by an enzyme defect; he also proved that Tay–Sachs patients could be diagnosed by an assay of hexosaminidase A activity.[45] The further development of enzyme assays demonstrated that levels of hexosaminidases A and B could be measured in patients and carriers, allowing the reliable detection of heterozygotes. During the early 1970s, researchers developed protocols for newborn testing, carrier screening, and pre-natal diagnosis.[30][46] By the end of 1979, researchers had identified three variant forms of GM2 gangliosidosis, including Sandhoff disease and the AB variant of GM2-gangliosidosis, accounting for false negatives in carrier testing.[47]
Society and culture[edit]
Since carrier testing for Tay–Sachs began in 1971, millions of Ashkenazi Jews have been screened as carriers. Jewish communities embraced the cause of genetic screening from the 1970s on. The success with Tay–Sachs disease has led Israel to become the first country that offers free genetic screening and counseling for all couples and opened discussions about the proper scope of genetic testing for other disorders in Israel.[48]
Because Tay–Sachs disease was one of the first autosomal recessive genetic disorders for which there was an enzyme assay test (prior to polymerase chain reaction testing methods), it was intensely studied as a model for all such diseases, and researchers sought evidence of a selective process. A continuing controversy is whether heterozygotes (carriers) have or had a selective advantage. The presence of four different lysosomal storage disorders in the Ashkenazi Jewish population suggests a past selective advantage for heterozygous carriers of these conditions.'[39]
This controversy among researchers has reflected three debates among geneticists at large:
- Dominance versus overdominance. In applied genetics (selective and agricultural breeding), this controversy has reflected the century-long debate over whether dominance or overdominance provides the best explanation for heterosis (hybrid vigor).
- The classical/balance controversy. The classical hypothesis of genetic variability, often associated with Hermann Muller, maintains that most genes are of a normal wild type, and that most individuals are homozygous for that wild type, while most selection is purifying selection that operates to eliminate deleterious alleles. The balancing hypothesis, often associated with Theodosius Dobzhansky, states that heterozygosity will be common at loci, and that it frequently reflects either directional selection or balancing selection.
- Selectionists versus neutralists. In theoretical population genetics, selectionists emphasize the primacy of natural selection as a determinant of evolution and of variation within a population, while neutralists favor a form of Motoo Kimura's neutral theory of molecular evolution, which emphasizes the role of genetic drift.[49]
Research directions[edit]
Enzyme replacement therapy[edit]
Enzyme replacement therapy techniques have been investigated for lysosomal storage disorders, and could potentially be used to treat Tay–Sachs as well. The goal would be to replace the nonfunctional enzyme, a process similar to insulin injections for diabetes. However, in previous studies, the HEXA enzyme itself has been thought to be too large to pass through the specialized cell layer in the blood vessels that forms the blood–brain barrier in humans.
Researchers have also tried directly instilling the deficient enzyme hexosaminidase A into the cerebrospinal fluid (CSF) which bathes the brain. However, intracerebral neurons seem unable to take up this physically large molecule efficiently even when it is directly by them. Therefore, this approach to treatment of Tay–Sachs disease has also been ineffective so far.[50]
Jacob sheep model[edit]
Tay–Sachs disease exists in Jacob sheep.[51] The biochemical mechanism for this disease in the Jacob sheep is virtually identical to that in humans, wherein diminished activity of hexosaminidase A results in increased concentrations of GM2 ganglioside in the affected animal.[52] Sequencing of the HEXA gene cDNA of affected Jacobs sheep reveal an identical number of nucleotides and exons as in the human HEXA gene, and 86% nucleotide sequence identity.[51] A missense mutation (G444R)[53] was found in the HEXA cDNA of the affected sheep. This mutation is a single nucleotide change at the end of exon 11, resulting in that exon's deletion (before translation) via splicing. The Tay–Sachs model provided by the Jacob sheep is the first to offer promise as a means for gene therapy clinical trials, which may prove useful for disease treatment in humans.[51]
Substrate reduction therapy[edit]
Other experimental methods being researched involve substrate reduction therapy, which attempts to use alternative enzymes to increase the brain's catabolism of GM2 gangliosides to a point where residual degradative activity is sufficient to prevent substrate accumulation.[54][55] One experiment has demonstrated that using the enzyme sialidase allows the genetic defect to be effectively bypassed, and as a consequence, GM2 gangliosides are metabolized so that their levels become almost inconsequential. If a safe pharmacological treatment can be developed – one that increases expression of lysosomal sialidase in neurons without other toxicity – then this new form of therapy could essentially cure the disease.[56]
Another metabolic therapy under investigation for Tay–Sachs disease uses miglustat.[57] This drug is a reversible inhibitor of the enzyme glucosylceramide synthase, which catalyzes the first step in synthesizing glucose-based glycosphingolipids like GM2 ganglioside.[58]
Increasing β-hexosaminidase A activity[edit]
As Tay–Sachs disease is a deficiency of β-hexosaminidase A, by getting a substance that increases its activity, people affected will not be deteriorating as fast or not at all. While for infantile Tay–Sachs disease, there is no β-hexosaminidase A so then the treatment would be ineffective. However, for people affected by Late-Onset Tay–Sachs disease, they still have β-hexosaminidase A. The drug pyrimethamine has been shown to increase activity of β-hexosaminidase A.[59] However, the increased levels of β-hexosaminidase A still fall far short of the desired '10% of normal HEXA', above which the phenotypic symptoms begin to disappear.[59]
Cord blood transplant[edit]
This is a highly invasive procedure which involves destroying the patient's blood system with chemotherapy and administering cord blood. Of five people who had received the treatment as of 2008, two were still alive after five years and they still had a great deal of health problems.[60]
Critics point to its harsh nature, and that it is unapproved. It is also hard for the blood to cross the blood–brain barrier, as well as very expensive, as each unit of cord blood costs $25,000 and adults need many units of cord blood.[61]
References[edit]
- ^ abcdefghijklmnopqr'Tay–Sachs disease'. Genetics Home Reference. October 2012. Archived from the original on 13 May 2017. Retrieved 29 May 2017.
- ^ abcdefghijkl'Tay Sachs Disease'. NORD (National Organization for Rare Disorders). 2017. Archived from the original on 20 February 2017. Retrieved 29 May 2017.
- ^Marinetti, G. V. (2012). Disorders of Lipid Metabolism. Springer Science & Business Media. p. 205. ISBN9781461595649. Archived from the original on 2017-11-05.
- ^Walker, Julie (2007). Tay–Sachs Disease. The Rosen Publishing Group. p. 53. ISBN9781404206977. Archived from the original on 2017-11-05.
- ^Vogel, Friedrich; Motulsky, Arno G. (2013). Vogel and Motulsky's Human Genetics: Problems and Approaches (3 ed.). Springer Science & Business Media. p. 578. ISBN9783662033562. Archived from the original on 2017-11-05.
- ^ abcde'Tay–Sachs disease Information Page'. National Institute of Neurological Disorders and Stroke. 14 February 2007. Archived from the original on 29 December 2011. Retrieved 10 May 2007.
- ^ abMcKusick, Victor A; Hamosh, Ada. 'Online Mendelian Inheritance in Man'. United States National Institutes of Health. Archived from the original on 29 December 2011. Retrieved 24 April 2009.
- ^Specola N, Vanier MT, Goutières F, Mikol J, Aicardi J (1 January 1990). 'The juvenile and chronic forms of GM2 gangliosidosis: clinical and enzymatic heterogeneity'. Neurology. 40 (1): 145–150. doi:10.1212/wnl.40.1.145. PMID2136940.
- ^Moe, P G; Benke, T A (2005). 'Neurologic and Muscular Disorders'. Current Pediatric Diagnosis and Treatment (17 ed.). McGraw-Hill. ISBN978-0-07-142960-3.
- ^Rosebush PI, MacQueen GM, Clarke JT, Callahan JW, Strasberg PM, Mazurek MF (1995). 'Late-onset Tay–Sachs disease presenting as catatonic schizophrenia: Diagnostic and treatment issues'. Journal of Clinical Psychiatry. 56 (8): 347–53. PMID7635850.
- ^Willner JP, Grabowski GA, Gordon RE, Bender AN, Desnick RJ (July 1981). 'Chronic GM2 gangliosidosis masquerading as atypical Friedreich's ataxia: Clinical, morphologic, and biochemical studies of nine cases'. Neurology. 31 (7): 787–98. doi:10.1212/wnl.31.7.787. PMID6454083.
- ^Kaback MM (December 2000). 'Population-based genetic screening for reproductive counseling: the Tay–Sachs disease model'. European Journal of Pediatrics. 159 (Suppl 3): S192–S195. doi:10.1007/PL00014401. ISSN1432-1076. PMID11216898.
- ^Myerowitz R (1997). 'Tay–Sachs disease-causing mutations and neutral polymorphisms in the Hex A gene'. Human Mutation. 9 (3): 195–208. doi:10.1002/(SICI)1098-1004(1997)9:3<195::AID-HUMU1>3.0.CO;2-7. PMID9090523.
- ^Myerowitz R, Costigan FC (15 December 1988). 'The major defect in Ashkenazi Jews with Tay–Sachs disease is an insertion in the gene for the alpha-chain of beta-hexosaminidase'. Journal of Biological Chemistry. 263 (35): 18587–18589. PMID2848800. Archived from the original on 17 April 2014.
- ^McDowell GA, Mules EH, Fabacher P, Shapira E, Blitzer MG (1992). 'The presence of two different infantile Tay–Sachs disease mutations in a Cajun population'. American Journal of Human Genetics. 51 (5): 1071–1077. PMC1682822. PMID1307230.
- ^Keats BJ, Elston RC, Andermann E (1987). 'Pedigree discriminant analysis of two French Canadian Tay–Sachs families'. Genetic Epidemiology. 4 (2): 77–85. doi:10.1002/gepi.1370040203. PMID2953646.
- ^De Braekeleer M, Hechtman P, Andermann E, Kaplan F (April 1992). 'The French Canadian Tay–Sachs disease deletion mutation: Identification of probable founders'. Human Genetics. 89 (1): 83–87. doi:10.1007/BF00207048. PMID1577470.
- ^Ohno K, Suzuki K (5 December 1988). 'Multiple Abnormal beta-Hexosaminidase Alpha-Chain mRNAs in a Compound-Heterozygous Ashkenazi Jewish Patient with Tay–Sachs Disease'(PDF). Journal of Biological Chemistry. 263 (34): 18563–7. PMID2973464. Archived(PDF) from the original on 26 September 2007. Retrieved 11 May 2007.
- ^ abKaback MM, Desnick RJ (2011). 'Hexosaminidase A Deficiency'. In Pagon RA, Adam MP, Ardinger HH, Bird TD, Dolan CR, Fong CT, Smith RJ, Stephens K (eds.). GeneReviews [Internet]. Seattle, Washington, USA: University of Washington, Seattle. PMID20301397. Archived from the original on 2014-01-16.
- ^Korf, Bruce R (2000). Human genetics: A problem-based approach (2 ed.). Wiley-Blackwell. pp. 11–12. ISBN978-0-632-04425-2.
- ^Mahuran DJ (1999). 'Biochemical consequences of mutations causing the GM2 gangliosidoses'. Biochimica et Biophysica Acta. 1455 (2–3): 105–138. doi:10.1016/S0925-4439(99)00074-5. PMID10571007.
- ^Hechtman P, Kaplan F (1993). 'Tay–Sachs disease screening and diagnosis: Evolving technologies'. DNA and Cell Biology. 12 (8): 651–665. doi:10.1089/dna.1993.12.651. PMID8397824.
- ^Tittarelli R, Giagheddu M, Spadetta V (July 1966). 'Typical ophthalmoscopic picture of 'cherry-red spot' in an adult with the myoclonic syndrome'. The British Journal of Ophthalmology. 50 (7): 414–420. doi:10.1136/bjo.50.7.414. PMC506244. PMID5947589.
- ^Aragão RE, Ramos RM, Pereira FB, Bezerra AF, Fernandes DN (Jul–Aug 2009). ''Cherry red spot' in a patient with Tay–Sachs disease: case report'. Arq Bras Oftalmol. 72 (4): 537–9. doi:10.1590/S0004-27492009000400019. PMID19820796.
- ^Seshadri R, Christopher R, Arvinda HR (2011). 'Teaching NeuroImages: MRI in infantile Sandhoff disease'. Neurology. 77 (5): e34. doi:10.1212/WNL.0b013e318227b215. PMID21810694.
- ^Stoller D (1997). 'Prenatal Genetic Screening: The Enigma of Selective Abortion'. Journal of Law and Health. 12 (1): 121–140. PMID10182027.
- ^'Chorionic Villus Sampling and Amniocentesis: Recommendations for Prenatal Counseling'. United States, Center for Disease Control. Archived from the original on 14 July 2009. Retrieved 18 June 2009.
- ^Bodurtha J, Strauss JF (2012). 'Genomics and perinatal care'. N. Engl. J. Med. 366 (1): 64–73. doi:10.1056/NEJMra1105043. PMC4877696. PMID22216843.
- ^Marik, J J (13 April 2005). 'Preimplantation Genetic Diagnosis'. eMedicine.com. Archived from the original on 31 January 2009. Retrieved 10 May 2007.
- ^ abEkstein, J; Katzenstein, H (2001). 'The Dor Yeshorim story: Community-based carrier screening for Tay–Sachs disease'. Tay–Sachs Disease. Advances in Genetics. 44. pp. 297–310. doi:10.1016/S0065-2660(01)44087-9. ISBN978-0-12-017644-1. PMID11596991.
- ^ abColaianni A, Chandrasekharan S, Cook-Deegan R (2010). 'Impact of Gene Patents and Licensing Practices on Access to Genetic Testing and Carrier Screening for Tay–Sachs and Canavan Disease'. Genetics in Medicine. 12 (4 Suppl): S5–S14. doi:10.1097/GIM.0b013e3181d5a669. PMC3042321. PMID20393311.
- ^Eeg-Olofsson L, Kristensson K, Sourander P, Svennerholm L (1966). 'Tay–Sachs disease. A generalized metabolic disorder'. Acta Paediatrica Scandinavica. 55 (6): 546–62. doi:10.1111/j.1651-2227.1966.tb15254.x. PMID5972561.
- ^Shapiro BE, Hatters-Friedman S, Fernandes-Filho JA, Anthony K, Natowicz MR (12 September 2006). 'Late-onset Tay–Sachs disease: Adverse effects of medications and implications for treatment'. Neurology. 67 (5): 875–877. doi:10.1212/01.wnl.0000233847.72349.b6. PMID16966555.
- ^Rozenberg R, Pereira Lda V (2001). 'The frequency of Tay–Sachs disease causing mutations in the Brazilian Jewish population justifies a carrier screening program'. Sao Paulo medical journal [Revista paulista de medicina]. 119 (4): 146–149. doi:10.1590/s1516-31802001000400007. PMID11500789.
- ^GM2 Gangliosidoses – Introduction And EpidemiologyArchived 2012-04-20 at the Wayback Machine at Medscape. Author: David H Tegay. Updated: Mar 9, 2012
- ^ abChakravarti A, Chakraborty R (1978). 'Elevated frequency of Tay–Sachs disease among Ashkenazic Jews unlikely by genetic drift alone'. American Journal of Human Genetics. 30 (3): 256–261. PMC1685578. PMID677122.
- ^ abcdFrisch A, Colombo R, Michaelovsky E, Karpati M, Goldman B, Peleg L (March 2004). 'Origin and spread of the 1278insTATC mutation causing Tay–Sachs disease in Ashkenazi Jews: Genetic drift as a robust and parsimonious hypothesis'. Human Genetics. 114 (4): 366–376. doi:10.1007/s00439-003-1072-8. PMID14727180.
- ^Koeslag JH, Schach SR (1984). 'Tay–Sachs disease and the role of reproductive compensation in the maintenance of ethnic variations in the incidence of autosomal recessive disease'. Annals of Human Genetics. 48 (3): 275–281. doi:10.1111/j.1469-1809.1984.tb01025.x. PMID6465844.
- ^ abRisch N, Tang H, Katzenstein H, Ekstein J (2003). 'Geographic Distribution of Disease Mutations in the Ashkenazi Jewish Population Supports Genetic Drift over Selection'. American Journal of Human Genetics. 72 (4): 812–822. doi:10.1086/373882. PMC1180346. PMID12612865.
- ^Slatkin M (2004). 'A Population-Genetic Test of Founder Effects and Implications for Ashkenazi Jewish Diseases'. American Journal of Human Genetics. 75 (2): 282–293. doi:10.1086/423146. PMC1216062. PMID15208782.
- ^Tay, Waren (1881). 'Symmetrical changes in the region of the yellow spot in each eye of an infant'. Transactions of the Ophthalmological Society. 1: 55–57.
- ^Sachs, Bernard (1887). 'On arrested cerebral development with special reference to cortical pathology'. Journal of Nervous and Mental Disease. 14 (9): 541–554. doi:10.1097/00005053-188714090-00001. hdl:10192/32703.
- ^ abReuter, Shelley Z (Summer 2006). 'The Genuine Jewish Type: Racial Ideology and Anti-Immigrationism in Early Medical Writing about Tay–Sachs Disease'. The Canadian Journal of Sociology. 31 (3): 291–323. doi:10.1353/cjs.2006.0061.
- ^'Amaurotic Idiocy'. The Jewish Encyclopedia. New York: Funk and Wagnalls. 1901–1906. Archived from the original on 29 December 2011. Retrieved 7 March 2009.
- ^Okada S, O'Brien JS (1969). 'Tay–Sachs disease: Generalized absence of a beta-D-N-acetylhexosaminidase component'. Science. 165 (3894): 698–700. Bibcode:1969Sci...165..698O. doi:10.1126/science.165.3894.698. PMID5793973.
- ^O'Brien JS, Okada S, Chen A, Fillerup DL (1970). 'Tay–Sachs disease: Detection of heterozygotes and homozygotes by serum hexaminidase assay'. New England Journal of Medicine. 283 (1): 15–20. doi:10.1056/NEJM197007022830104. PMID4986776.
- ^O'Brien, John S (1983). 'The Gangliosidoses'. In Stanbury, J B; et al. (eds.). The Metabolic Basis of Inherited Disease. New York: McGraw Hill. pp. 945–969.
- ^Sagi M (1998). 'Ethical aspects of genetic screening in Israel'. Science in Context. 11 (3–4): 419–429. doi:10.1017/s0269889700003112. PMID15168671.
- ^Kimura, Motoo (1983). The Neutral Theory of Molecular Evolution. Cambridge: Cambridge University Press. ISBN978-0-521-23109-1.
- ^Matsuoka K, Tamura T, Tsuji D, Dohzono Y, Kitakaze K, Ohno K, Saito S, Sakuraba H, Itoh K (14 October 2011). 'Therapeutic Potential of Intracerebroventricular Replacement of Modified Human β-Hexosaminidase B for GM2 Gangliosidosis'. Molecular Therapy. 19 (6): 1017–1024. doi:10.1038/mt.2011.27. PMC3129794. PMID21487393. Archived from the original on 21 August 2014.
- ^ abcTorres PA, Zeng BJ, Porter BF, Alroy J, Horak F, Horak J, Kolodny EH (2010). 'Tay–Sachs disease in Jacob sheep'. Molecular Genetics and Metabolism. 101 (4): 357–363. doi:10.1016/j.ymgme.2010.08.006. ISSN1096-7192. PMID20817517.
- ^Porter BF, Lewis BC, Edwards JF, Alroy J, Zeng BJ, Torres PA, Bretzlaff KN, Kolodny EH (2011). 'Pathology of GM2 Gangliosidosis in Jacob Sheep'. Veterinary Pathology. 48 (3): 807–813. doi:10.1177/0300985810388522. ISSN0300-9858. PMID21123862.
- ^Kolodny E, Horak F, Horak J (2011). 'Jacob sheep breeders find more Tay–Sachs carriers'. ALBC Newsletter. Archived from the original on 29 December 2011. Retrieved 5 May 2011.
- ^Platt FM, Neises GR, Reinkensmeier G, Townsend MJ, Perry VH, Proia RL, Winchester B, Dwek RA, Butters TD (1997). 'Prevention of lysosomal storage in Tay–Sachs mice treated with N-butyldeoxynojirimycin'. Science. 276 (5311): 428–431. doi:10.1126/science.276.5311.428. PMID9103204.
- ^Lachmann RH, Platt FM (2001). 'Substrate reduction therapy for glycosphingolipid storage disorders'. Expert Opinion on Investigational Drugs. 10 (3): 455–466. doi:10.1517/13543784.10.3.455. PMID11227045.
- ^Igdoura SA, Mertineit C, Trasler JM, Gravel RA (1999). 'Sialidase-mediated depletion of GM2 ganglioside in Tay–Sachs neuroglia cells'. Human Molecular Genetics. 8 (6): 1111–1116. doi:10.1093/hmg/8.6.1111. PMID10332044.
- ^'Pharmacokinetics, Safety and Tolerability of Zavesca (Miglustat) in Patients With Infantile Onset Gangliosidosis: Single and Steady State Oral Doses'. 5 May 2008. Archived from the original on 13 February 2012. Retrieved 10 April 2012.
- ^Kolodny EH, Neudorfer O, Gianutsos J, Zaroff C, Barnett N, Zeng BJ, Raghavan S, Torres P, Pastores GM (2004). 'Late-onset Tay–Sachs disease: Natural history and treatment with OGT 918 (Zavesca™)'. Journal of Neurochemistry. 90 (S1): 54–55. Bibcode:2006JNeur..26.9606G. doi:10.1111/j.1471-4159.2004.02650_.x. ISSN0022-3042.
- ^ abOsher E, Fattal-Valevski A, Sagie L, Urshanski N, Amir-Levi Y, Katzburg S, Peleg L, Lerman-Sagie T, Zimran A, Elstein D, Navon R, Stern N, Valevski A (March 2011). 'Pyrimethamine increases β-hexosaminidase A activity in patients with Late Onset Tay Sachs'. Mol. Genet. Metab. 102 (3): 356–63. doi:10.1016/j.ymgme.2010.11.163. PMID21185210.
- ^Prasad, Vinod K.; Mendizabal, Adam; Parikh, Suhag H.; Szabolcs, Paul; Driscoll, Timothy A.; Page, Kristin; Lakshminarayanan, Sonali; Allison, June; Wood, Susan (2008-10-01). 'Unrelated donor umbilical cord blood transplantation for inherited metabolic disorders in 159 pediatric patients from a single center: influence of cellular composition of the graft on transplantation outcomes'. Blood. 112 (7): 2979–2989. doi:10.1182/blood-2008-03-140830. ISSN0006-4971. PMC2556628. PMID18587012.
- ^William Hathaway (May 16, 2006). 'Umbilical Cord Blood Is Child's Last Hope, Stem Cells Could Halt Tay–Sachs Damage'. Hartford Courant.
External links[edit]
| Classification |
|
|---|---|
| External resources |
|
| Wikimedia Commons has media related to Tay–Sachs disease. |
- Tay–Sachs disease at NLM Genetics Home Reference
Retrieved from 'https://en.wikipedia.org/w/index.php?title=Tay–Sachs_disease&oldid=894645247'
 RSS Feed
RSS Feed
